|
The uptake of aluminum can take place through food, through breathing and by skin contact. They are usually found in a solution of aluminum in combination with other ions, for instance as aluminum chlorine. The water-soluble form of aluminum causes the harmful effects, these particles are called ions. But still, when one is exposed to high concentrations, it can cause health problems. Due to these facts, aluminum is commonly known as an innocent compound. 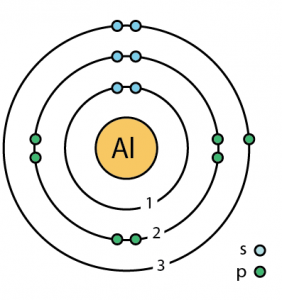
Health effects of aluminumĪluminum is one of the most widely used metals and also one of the most frequently found compounds in the earth's crust. Known reserves of ores are 6 billion tones. Industrial production world-wide of new metal is around 20 million tons per year, and a similar amount is recycled. Recovery of this metal from scrap (via recycling) has become an important component of the aluminum industry. Topaz is aluminum silicate coloured yellow by traces of iron. Both these are now easy and cheap to manufacture artificially. The presence of traces of other metals creates various colors: cobalt creates blues sapphires, and chromium makes red rubies. Several gemstones are made of the clear crystal form of aluminum oxide known as corundum. The very reason for which aluminum is used in many applications is why it is so hard to produce. Aluminum is among the most difficult metals on earth to refine, the reason is that aluminum is oxidized very rapidly and that its oxide is an extremely stable compound that, unlike rust on iron, does not flake off. 
Aluminum contribute greatly to the properties of soil, where it is present mainly as insoluble aluminum hydroxide.Īluminum is a reactive metal and it is hard to extract it from its ore, aluminum oxide (Al 2O 3). The result is a solid foam which is used in traffic tunnels and in space shuttle.Īluminum is an abundant element in Earth's crust: it is believed to be contained in a percentage from 7.5% to 8.1%. The molten aluminum has to he thickened before this is done and this is achieved by adding aluminum oxide or silicon carbide fibers. The most recent development in aluminum technology is the production of aluminum foam by adding to the molten metal a compound (a metal hybrid), which releases hydrogen gas. Unfortunately problems on the functioning were caused by its greater coefficient of thermal expansion and its tendency to creep under steady sustained pressure, both eventually causing loosening the connection galvanic corrosion increasing the electrical resistance. 
Other applications are electrical transmission lines, and packaging (cans, foil, etc.).īecause of its high conductivity and relatively low price compared to copper, aluminum was introduced for household electrical wiring to a large degree in the US in the 1960s. Telescope mirrors are also coated with a thin layer of aluminum. Nearly all modern mirrors are made using a thin reflective coating of aluminum on the back surface of a sheet of float glass. Pure aluminum easily forms alloys with many elements such as copper, zinc, magnesium, manganese and silicon. The use of aluminum exceed that of any other metal except iron. Structural components made from aluminum are vital to the aerospace industry and very important in other areas of transportation and building in which light weight, durability, and strength are needed. Aluminum is used in many industries to make millions of different products and is very important to the world economy. Aluminum is nontoxic (as the metal) nonmagnetic and non-sparking.Īluminum has only one naturally occurring isotope, aluminium-27, which is not radioactive.Ī silvery and ductile member of the poor metal group of elements, aluminum is found primarily as the ore bauxite and is remarkable for its resistance to oxidation (aluminum is actually almost always already oxidized, but is usable in this form unlike most metals), its strength, and its light weight. It has a dull silvery appearance, because of a thin layer of oxidation that forms quickly when it is exposed to air. However, in the USA the preferred name was aluminum and when the American Chemical Society debated on the issue, in 1925, it decided to stick with aluminum.Īluminum is a soft and lightweight metal. Aluminum was the original name given to the element by Humphry Davy but others called it aluminum and that became the accepted name in Europe. 
The name aluminum is derived from the ancient name for alum (potassium aluminum sulphate), which was alumen (Latin, meaning bitter salt). Separation and Concentration Purification RequestĪluminum - Al Properties - Health effects of aluminum - Environmental effects of aluminum.Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
